Clinical Study on Reflusod for Infant Reflux Relief
Infant gastroesophageal reflux (GER) is a common condition in newborns and infants, often marked by frequent regurgitation, inconsolable crying, irritability, feeding difficulties, and disrupted sleep. According to the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), up to 50% of healthy infants may experience reflux symptoms in the first months of life.
Additionally, the MSD Manual — one of the most widely referenced clinical resources by pediatricians — notes that infant GER is often benign but may require treatment when associated with discomfort or failure to thrive.
If persistent, these symptoms can significantly affect the infant’s well-being and lead to parental stress.
Reflusod is a CE-marked oral medical device specifically formulated to safely and effectively manage infant reflux symptoms. Its action is based on the formation of a protective gastroesophageal barrier, helping to limit mechanical and acidic irritation while promoting digestive comfort.
The clinical data presented are derived from a prospective observational study conducted by Farmaimpresa Group in May 2025.
💡 To learn more about the regulatory and clinical context of GERD devices, read our article on how GFI has extended certificates for GERD medical devices, ensuring the regulatory reliability of solutions like Reflusod.
Clinical Study on Reflusod in Infants with GER: Methodology & Outcomes
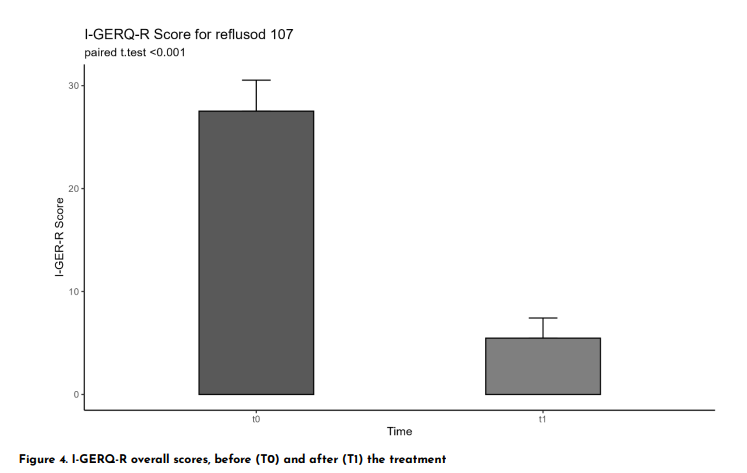
An observational clinical study was conducted on 20 infants under 12 months of age with symptoms consistent with gastroesophageal reflux (GER). Evaluation was performed using the validated I-GERQ-R (Infant Gastroesophageal Reflux Questionnaire Revised) before (T0) and after 4 weeks of treatment with Reflusod (T1).
The questionnaire measured parameters such as:
- Frequency of regurgitation
- Crying associated with feeding
- Postprandial irritability
- Sleep disturbances and feeding difficulties
Clinical Improvements: 75% Reduction in GER Symptoms
After 4 weeks of treatment, the average I-GERQ-R score dropped from 21.7 to 10.9 points, with a mean reduction of 10.8 points (p < 0.001).
- 85% of infants showed clinically significant improvement (≥5-point reduction)
- 75% of cases achieved a ≥50% reduction in symptoms
This data confirms Reflusod as an effective solution for rapid improvement of key reflux symptoms, positively impacting infant behavior, sleep, and feeding patterns.
Want to compare other approaches to infant reflux?
For a broader overview of drugs, medical devices, and anti-reflux treatments, please refer to our dedicated article on anti-reflux preparations: a comparison between drugs and medical devices. A valuable resource for physicians, distributors, and healthcare professionals.
Looking for a pediatric line ready for distribution?
We are certified manufacturers of medical devices based on substances, dietary supplements, and cosmetics. Reflusod is ready for market entry or private labeling, with full regulatory, marketing, and clinical support.