A Medical Device Based on Sodium Hyaluronate to Create the Ideal Environment for Natural Conception
I am text block. Click edit button to change this text. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
The Key Role of Sperm Motility in Conception
Sperm motility is the ability of spermatozoa to move autonomously through the female reproductive tract to reach and fertilize the ovum. Factors such as vaginal pH, osmolality, and the presence of nutrients can drastically influence the vitality and progression of spermatozoa. An unsuitable environment can significantly reduce their ability to move, hindering the path to fertilization. For this reason, any product intended for intimate use in the context of seeking pregnancy must demonstrate that it does not negatively interfere with this vital function.
We conducted an in-depth clinical study on our product FT.CE.107, an innovative single-dose gel treatment. This medical device is specifically formulated to help couples facing infertility (WHO), aiming to optimize conditions for natural conception.
Our medical device is based on a carefully balanced formulation that includes:
- Sodium Hyaluronate
- Creatine
- Carnitine
- Inositol
- Calcium Pyruvate
XY AQUAGEL: An Optimal Environment for Spermatozoa
XY AQUAGEL is a medical device formulated for fertility support, with a specific action designed to:
- Create an optimal environment for spermatozoa.
- Help maintain a suitable pH for natural conception.
- Be characterized by a seminal fluid-compatible osmolality.
- Mimic seminal fluid composition, thereby generating a comfortable environment for spermatozoa.
- Provide adequate energy for sperm motility.
These characteristics are fundamental to ensure that spermatozoa maintain their vitality and their ability to move on the path to the ovum.
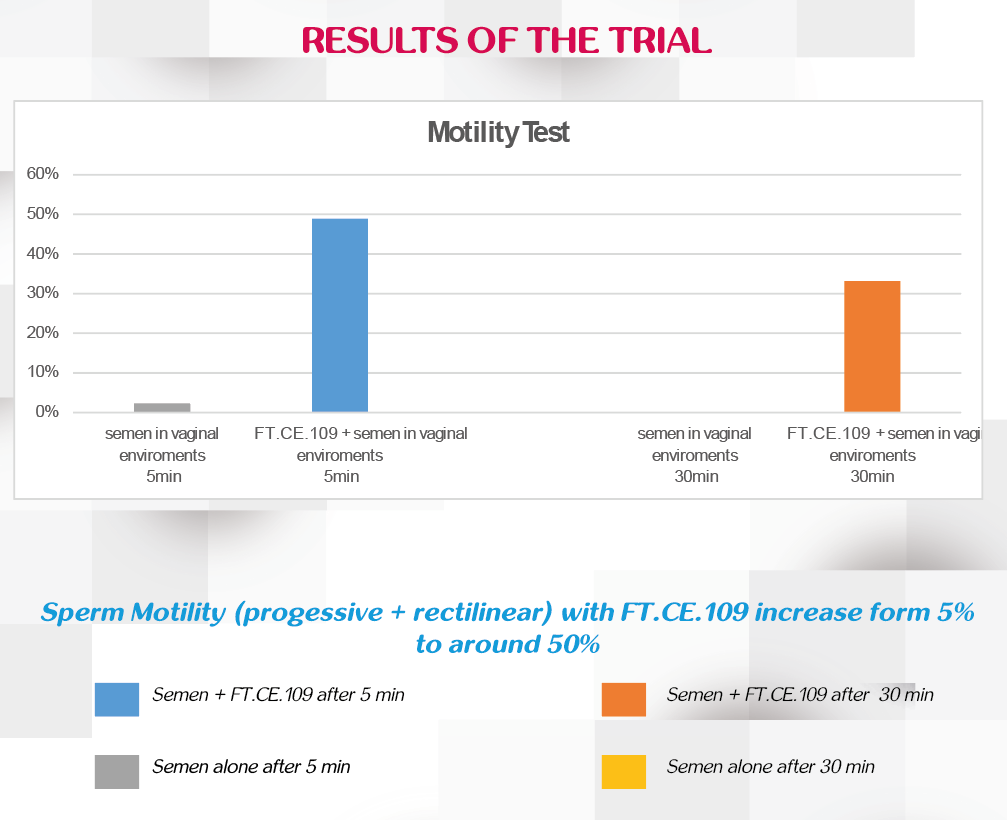
The Motility Test: Validation of XY AQUAGEL Efficacy
To confirm that it effectively meets its intended use and supports sperm function, a clinical study was conducted as part of the evaluation protocol. This ex vivo test was essential to measure the product’s impact on the ability of spermatozoa to move effectively and progressively. By evaluating sperm motility in the presence of our vaginal gel, it was shown that the medical device is not only biocompatible but actively contributes to maintaining an environment favorable to sperm motility.
Through this evaluation, our medical device positions itself as an ally for couples seeking to conceive, offering an intimate environment that respects and supports the delicate phase of sperm motility.
If you are interested in becoming a distributor of our medical device XY AQUAGEL, a single-dose gel treatment based on sodium hyaluronate, CE certified in compliance with Regulation (UE) 2017/745 MDR (Medical Device Regulation), both in private label and out-licensing, we are ready to support your growth and provide personalized solutions for your market. Our attention to quality and customer satisfaction makes us the ideal partner to expand your offering in the health sector.