Clinical study on Reflusod in adults: significant improvement in GERD symptoms
Gastroesophageal reflux disease (GERD) is one of the most widespread digestive conditions in the adult population. Symptoms such as retrosternal burning, acid regurgitation, dysphagia, abdominal bloating, and sleep disturbances can significantly impair quality of life and work performance.
Gastroesophageal reflux in adults is internationally recognized as a condition with a high impact on quality of life. The guidelines of the World Gastroenterology Organisation (WGO) emphasize the importance of proper symptom evaluation and the use of validated tools to monitor treatment effectiveness.
The MSD Manual – Professional Version also highlights how GERD can present in erosive and non-erosive forms (NERD), with symptoms such as retrosternal burning, regurgitation, dysphagia, and sleep disturbances, which can significantly affect the daily well-being of adult patients.
According to international scientific literature, GERD can manifest in different clinical forms: NERD (Non-Erosive Reflux Disease), functional heartburn, hypersensitive esophagus to acid and non-acid reflux.
In this context comes Reflusod (FT.CE.107.H), an oral CE medical device developed by Gruppo FarmaImpresa for the treatment of symptoms related to gastroesophageal reflux in adults.
The reported data refer to an observational clinical study conducted by Gruppo Farmaimpresa in May 2025.
💡 To explore the regulatory and clinical context of GERD devices, also read the article on how GFI has extended certifications for GERD medical devices, ensuring the regulatory reliability of solutions such as Reflusod.
Clinical study on Reflusod in adults: methodology
The prospective observational clinical study involved:
- 20 adult patients
- Average age: 58.45 years
- Treatment duration: 4 weeks
- Assessment at baseline (T0) and post-treatment (T1)
Assessment tool: GERD-HRQL
Efficacy was measured using the GERD-HRQL (Gastroesophageal Reflux Disease – Health Related Quality of Life), an internationally validated questionnaire to assess:
- Intensity of heartburn
- Regurgitation
- Heartburn in supine position
- Impact on diet
- Night awakenings
- Dysphagia
- Abdominal bloating
- Patient satisfaction
Clinical results in adults: significant reduction in GERD-HRQL score
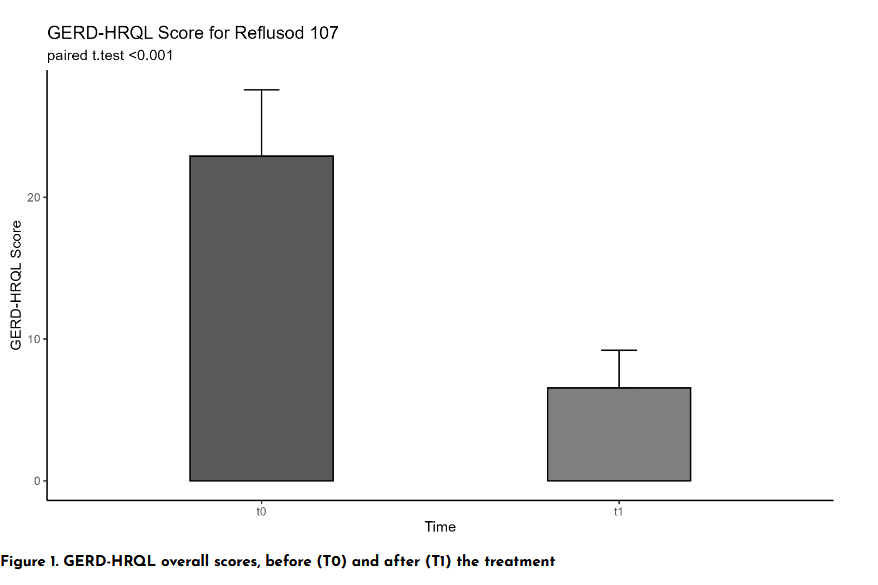
As illustrated in the graph of overall GERD-HRQL scores, the mean value dropped dramatically from 22.90 (T0) to 6.55 (T1) after just 4 weeks of treatment, with extremely strong statistical significance (p < 0.001).
This improvement is not only numerical: 100% of patients achieved at least a 50% reduction in score, reaching a threshold below 16 points which, according to the reference scientific literature, indicates clinical remission of GERD.
Analyzing individual parameters, a near-complete resolution of the most disabling symptoms is observed, such as nocturnal heartburn and postprandial regurgitation, restoring patients to optimal quality of life and lasting digestive comfort.
Mechanism of action
Reflusod acts by forming a protective gastroesophageal mechanical barrier, capable of:
- Limiting acid contact with the mucosa
- Reducing esophageal irritation
- Decreasing the frequency of regurgitation
- Improving digestive comfort
It does not interfere with gastric physiology but exerts a local protective action.
Who is Reflusod indicated for?
A clinically validated solution for:
- Gastroesophageal reflux disease (GERD)
- NERD
- Functional heartburn
- Hypersensitive esophagus to acid and non-acid reflux
- Persistent uncontrolled symptoms
Suitable for gastroenterological settings and integrated management of adult patients.
Why choose Reflusod?
- Prospective clinical study in adults
- 71% reduction in GERD-HRQL score
- 100% of patients with ≥50% improvement
- Assessment using an internationally validated questionnaire
Looking for a ready-to-market B2B or private label solution?
We are certified manufacturers of medical devices, food supplements, and cosmetics. Reflusod (FT.CE.107.H) is ready for market launch or available in private label format, with full regulatory and clinical support.