Aleman: Clinical Study in the Treatment of Constipation in Parkinson’s Patients
Constipation is one of the most frequent non-motor symptoms in patients suffering from Parkinson’s disease, with a prevalence that can reach 80%. It often precedes motor symptoms by years, contributing significantly to a worsening of the quality of life. In addition to the dysfunction of enteric innervation caused by alpha-synuclein aggregates, dopaminergic drugs can also worsen the intestinal picture.
It is in this context that the study on Aleman, a medical device based on fructo-oligosaccharides (FOS) and D-mannitol, is designed to improve intestinal motility and counteract chronic constipation in Parkinson’s patients.
Clinical Study Method
The 6-week observational clinical study involved 20 patients diagnosed with Parkinson’s and constipation according to the Rome II criteria.
The Rome II criteria are international clinical guidelines used to diagnose functional gastrointestinal disorders, such as constipation and irritable bowel syndrome, based on the presence and frequency of symptoms.
All subjects discontinued their usual laxatives to start treatment with Aleman, dissolved in lukewarm water, in the morning. The dosage could vary from 1 to 3 sachets per day based on the response.
The following were evaluated:
- Weekly frequency of bowel movements
- Use of rescue rectal laxatives
- Score on a questionnaire about 4 symptoms: straining during evacuation, sensation of incomplete emptying, tenesmus, hard stools (total score from 0 to 12)
Clinical Results: Significant Improvements in Bowel Movements, Laxatives, and Quality of Life
The effectiveness of the treatment with Aleman was measured considering three fundamental parameters: average number of bowel movements, use of daily laxatives, and the score of the qualitative questionnaire administered to patients.
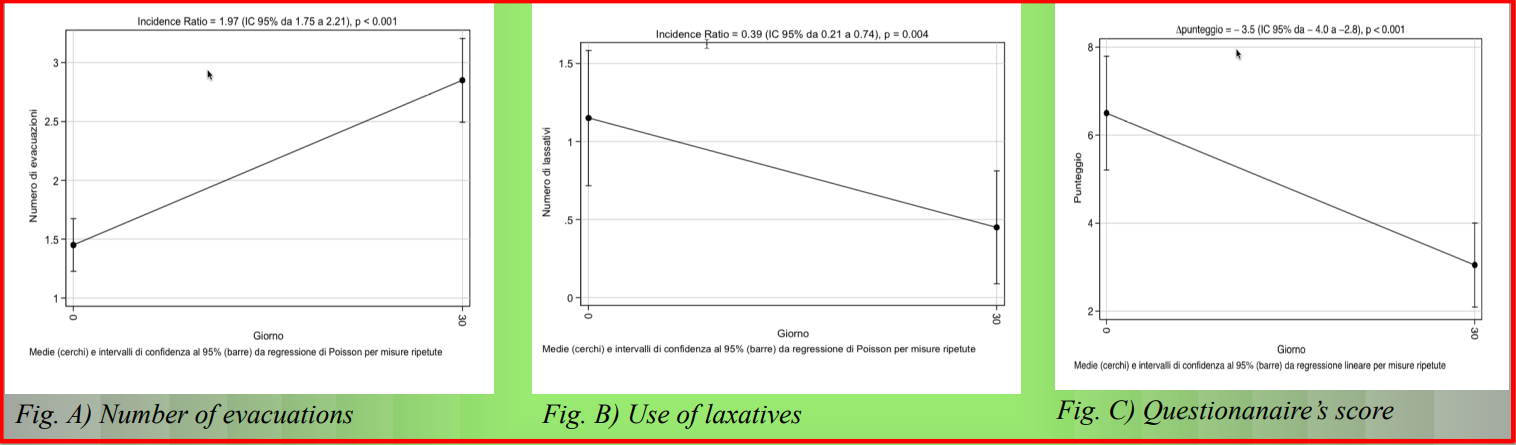
As illustrated in the following graph:
- Fig. A: The average number of bowel movements almost doubled after 30 days of treatment (IR = 1,97; IC 95%: 1,75–2,21; p < 0,001)
- Fig. B: The use of laxatives was drastically reduced (IR = 0,39; IC 95%: 0,21–0,74; p = 0,004)
- Fig. C: Subjective discomfort decreased in a statistically significant way (−3,5 points; IC 95%: −4,0 a −2,8; p < 0,001), signalling a clear improvement in the perception of intestinal well-being.
These results highlight the effectiveness of Aleman in promoting intestinal regularity, reducing the need for laxatives, and improving digestive comfort in patients in a short time, making it a safe and well-tolerated option for the management of constipation.
Conclusions
Aleman is confirmed as an effective and well-tolerated strategy for the management of constipation in patients suffering from Parkinson’s disease. Its synergistic combination acts on both stool consistency and intestinal motility, improving the quality of life and the absorption of medications.
Are you looking for a ready-to-use solution for B2B or private label distribution?
We are manufacturers of medical devices, food supplements, and cosmetics. Aleman is ready for market launch or private label, with full regulatory and clinical support.