The oral absorption of active ingredients (AIs) is influenced by several factors that determine the amount of AI that reaches the bloodstream.
These factors include the rate of dissolution in body fluids, the permeability of the gastrointestinal membrane, the release of the active ingredient from the pharmaceutical form, the rate of gastric emptying, and intestinal metabolism.
Ingested active ingredients pass through the gastrointestinal tract, starting from the esophagus with peristaltic movements that push the drug towards the stomach, where the residence time varies from 5 minutes to 2 hours, influenced by particle size.
In the small intestine, responsible for most nutrient absorption, the transit time is about 3-4 hours, essential for bioavailability, especially for extended-release formulations.
The colon, the final part of the gastrointestinal tract, has a variable transit time from 12 to 36 hours, where it absorbs water and substances, compacting the feces. These factors are crucial for the effectiveness of active ingredient absorption and must be considered in pharmaceutical formulation design to ensure adequate bioavailability.
Alpha-lipoic acid (ALA), known for its antioxidant properties, is widely used in clinical settings, but its effectiveness is limited by its bioavailability, only 30% due to high metabolism before reaching systemic circulation.
Despite being rapidly absorbed, ALA intake is influenced by the presence of food, which reduces its absorption. It crosses the blood-brain barrier and acts as a direct and indirect antioxidant, neutralizing reactive oxygen species and forming chelating complexes with transition metals.
ALA also induces the expression of the enzyme heme oxygenase 1 (HO-1), suggesting an additional mechanism of cellular protection, potentially anti-inflammatory. Improving the bioavailability of ALA is crucial to ensure optimal plasma concentrations and maximize its pharmacodynamic effects.
ALA absorption can vary with age and is not significantly affected by gastric emptying time.
However, its chemical instability and tendency to polymerize pose challenges for improving its bioavailability. Strategies to enhance absorption include using technologies that stabilize the active ingredient, ensuring a more effective and controlled release.
Physio Release® Technology
Physio Release® Technology represents a significant advancement in controlled-release systems for oral administration of active ingredients. Compared to traditional methods that involve the preparation of modified-release tablets, this technology combines the active ingredient with inert excipients in a compressed conglomerate. These excipients are specially selected to regulate the release of the active ingredient in a specific way:
- Rate: The release rate of the active ingredient can be slowed down to reduce the frequency of administrations.
- Time: The release can be delayed so that it starts after a certain period following intake.
- Site: The active ingredient can be released in a specific part of the gastrointestinal tract, such as the small intestine.
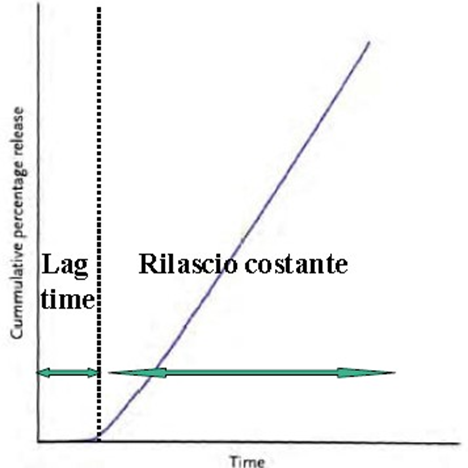
The primary goal of Physio Release® Technology is to ensure constant absorption over time, maintaining blood levels of the active ingredient within the desired therapeutic range. Additionally, it aims to make the active ingredient available for absorption only in areas where it can occur effectively, such as the small intestine, preventing its release and possible degradation in other parts of the gastrointestinal tract, like the colon.
Once taken on an empty stomach, the tablet begins to dissolve approximately 30 minutes after ingestion when it is in the small intestine. This dissolution process continues for about 4 hours, allowing the active ingredient to be fully released into the body fluids of the small intestine, ensuring it does not reach the colon where it would no longer be absorbed.
With Physio Release®, the time and site of active ingredient release can be adjusted to maximize therapeutic benefits
Bibliography:
Custodio, J.M., Wu Chi-Yuan, Benet, L. (2008) Predicting drug disposition, absorption/elimination/ transporter interplay and the role o ood on drug absorption.
Advanced Drug Delivery Reviews, 60, 717–733. Davit, B., Conner, D. (2008) Food effects on drug bioavailability: Implications or new and generic drug development. In: Krishna, R., Yu, L. (eds)
Biopharmaceutics Applications in Drug Development. Springer, New York.
Fleisher, D., Sweet, B.V., Parekh, A., Boullata, J.I. (2010) Drug absorption with Food. In: Handbook of Drug Nutrient Interactions, Nutrition and Health, Part 3, 209–241. Humana Press, Springer, New York.
Gray, V., Dressman, J. (1996) Change of pH requirements or simulated intestinal f uid TS. Pharmacopeial Forum, 22, 1943–1945.
Dobson, P.D., Kell, D.B. (2008) Carrier-mediated cellular uptake of pharmaceutical drugs: an exception or the rule? Nature Drug Discovery Reviews, 7, 205–220.
Hu, M., Li, X. (eds) (2011) Bioavailability: Basic Principles, Advanced Concepts, and Applications. John Wiley & Sons, New Jersey.
McConnell, E.L., Fadda, H.M., Basit, A.W. (2008) Gut instincts: Explorations in intestinal physiology and drug delivery. International Journal of Pharmaceutics, 34, 213–226.
Sugano, K., Kansy, M., Artursson, P., Avdee , A., Bendels,
S., Di, L., et al. (2010) Coexistence of passive and
carrier-mediated processes in transport. Nature Reviews Drug Discovery, 9, 597–614.
Rochette L, Ghibu S, Richard C, et al. Direct and indirect antioxidant properties of
α-lipoic acid and therapeutic potential. Mol Nutr Food Res 2013;57:114-25.
2 Teichert J, Hermann R, Ruus P, et al. Plasma kinetics, metabolism, and urinary excretion
of alpha-lipoic acid following oral administration in healthy volunteers. J Clin Pharmacol 2003;43:1257-67.
3 Teichert J, Tuemmers T, Achenbach H, et al. Pharmacokinetics of alpha-lipoic acid in subjects with severe kidney damage and end-stage renal disease. J Clin Pharmacol 2005;45:313-28.
4 Mignini F, Nasuti C, Gioventu G, et al. Human bioavailability and pharmacokinetic
profile of different formulations delivering alpha lipoic acid. Journal of Bioequivalence and Bioavailability 2012;1:1-6.
5 Keith DJ, Butler JA, Bemer B, et al. Age and gender dependent bioavailability of R- and R, S-α-lipoic acid: a pilot study. Pharmacol Res 2012;66:199-206.
6 Koufaki M. Therapeutic applications of lipoic acid: a patent review (2011 – 2014). Expert Opin Ther Pat 2014;24:993-1005.
7 Kisanuki A, Kimpara Y, Oikado Y, et al. Ring-opening polymerization of lipoic acid and
characterization of the polymer. Journal of Polymer Science Part A: Polymer Chemistry 2010;48:5247-53.
8 Carlson DA, Smith AR, Fischer SJ, et al. The plasma pharmacokinetics of R-(+)-lipoic acid administered as sodium R-(+)-lipoate to healthy human subjects. Altern Med Rev 2007;12:343-51.